Iron-based Hydrogen Storage
By making use of steam and iron pellets, hydrogen can be created with the only byproduct being rust and heat. By regenerating the rust with hydrogen, a completely circular, compact, safe and energy efficient hydrogen storage method is created.

Iron pellets
This article is a part of our University Technology Exposure Program. The program aims to recognize and reward innovation from engineering students and researchers across the globe.
Community voting is now live. Vote for your favorite submission by visiting: University Technology Exposure Program Community Vote.
That hydrogen has a lot of potential as a fuel has been known for years, the first mention of a hydrogen economy happened in the year 1970. Still, there is a long way to go to make hydrogen fit in our future energy mix. Hydrogen, when produced with renewable energy, is a sustainable energy carrier. However, hydrogen has some problems. It is a flammable and light gas and must be stored under high pressure. This makes transport and seasonal storage of hydrogen difficult and therefore expensive.
Iron-based Hydrogen Storage (IRHYS) offers an alternative to existing hydrogen storage options. Instead of storing hydrogen under high pressure, iron is used as a storage technique. We as SOLID are planning to have a proof of concept for generating hydrogen from iron at the beginning of this summer.
The Process
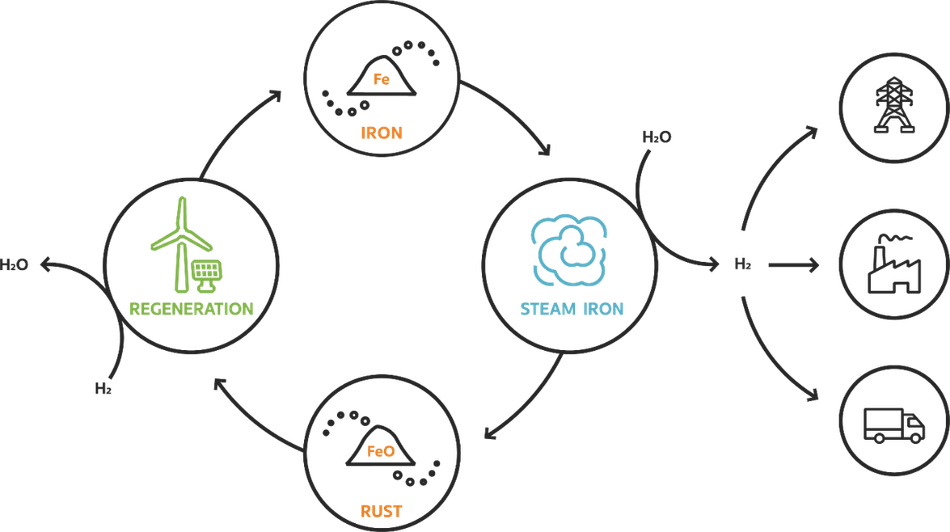
Oxidizing iron using steam (the steam-iron process) creates iron oxide (rust) and hydrogen. This hydrogen can be separated from the rust and can then be used for various applications. They produced iron oxide that can be regenerated by adding green hydrogen to the iron oxide. This creates a sustainable cycle where hydrogen can be stored and released when you need it. The cycle is shown in Figure 1.
IRHYS systems provide an efficient, safe, and possibly cheaper alternative to current hydrogen storage techniques. Iron is easier to store and transport than hydrogen because it only ignites at higher temperatures. Besides, it does not need to be stored under any pressure. Iron is a versatile substance. Not only is iron suitable for hydrogen storage, but the iron powder is fuel on its own.
In this project, SOLID is going to make a proof of concept of the steam-iron oxidation reaction (the right part of the total cycle in an IRHYS system). The main focus of the project is the realization of a steam-iron oxidation reactor. This reactor must be suitable for converting iron pellets into rust and hydrogen by letting it react with steam. The technology is expected to be highly scalable and can be used to replace current hydrogen storage
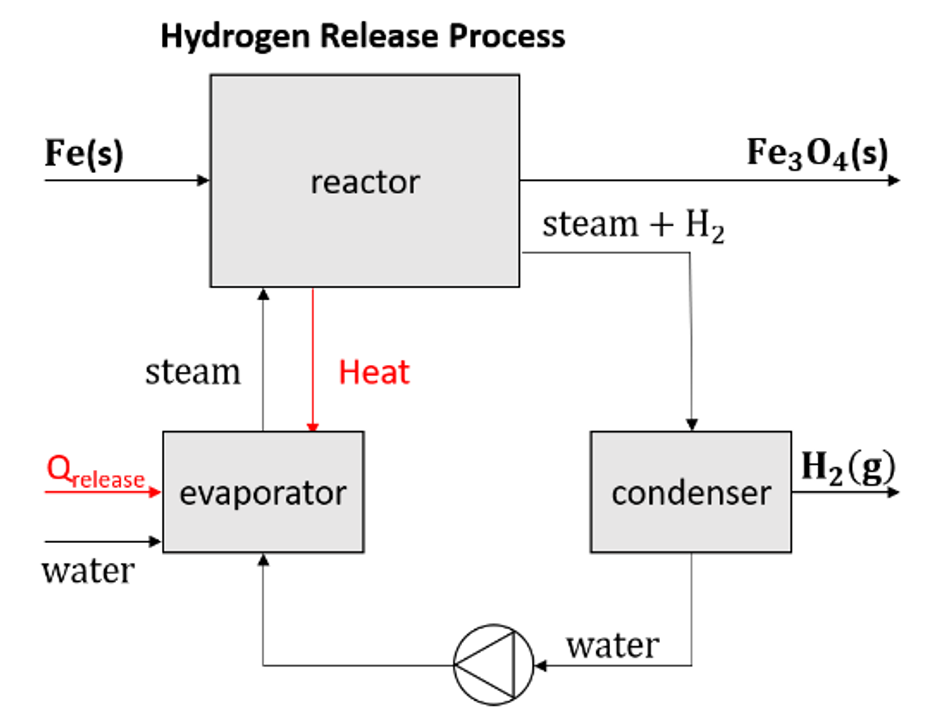
methods in various sectors. A simplified scheme of the oxidation process, which is taking place in the reactor, is shown in figure 2.
The process starts with placing the iron pellets in the steam-iron oxidation reactor. With the help of an evaporator, steam is added to the reactor. This causes the oxidation reaction to take place. This reaction is an exothermic reaction that releases heat that can be used in the evaporator to generate more steam. The reaction that takes place in the reactor is:
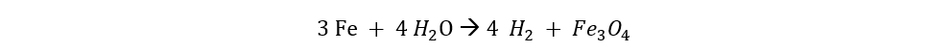
After this reaction has taken place there is still a mix of hydrogen and water (steam) left, the water is filtered out in the condenser so that pure hydrogen is left and can be extracted from the reactor.
An obstacle to our innovation is the lack of information about the reactor. As there is no physical reactor yet, it is not known what the exact purity of the created hydrogen, the total amount of hydrogen produced, or the reaction kinetics will be. These information gaps will be found during the testing phase of the proof of concept. This will take place in May 2022.
Application
Currently, a lot of countries are building wind turbines and placing solar panels to achieve the goals described in the climate agreement and gain more green energy. However, the sun does not always shine and the wind does not always blow. While on the other hand there are moments that the energy supply is higher than the energy demand. The latter is the moment when the rust has to be regenerated to iron pellets. Whereafter these can be transported to anywhere in the world. Where the hydrogen can be extracted by oxidizing the pellets.
Industries that use hydrogen as a fuel can use iron to store this hydrogen without having to keep it under strict storage regulations. Furthermore, transportation of hydrogen in iron can be done through the current infrastructure, so there are no changes required there.
Iron Fuel
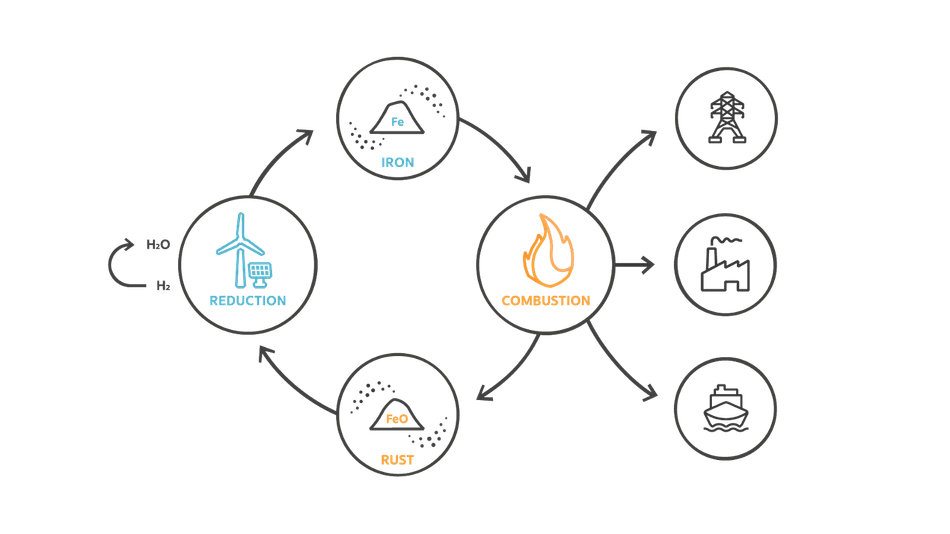
Iron has an additional function, it can also be used as fuel to generate heat. The process has a similar cycle as the IRHYS technology, shown in figure 3.
In this technology, iron can be used as a method of storing energy in general, instead of merely hydrogen. The iron will be combusted instead of oxidized with steam, generating much heat. Iron Fuel has already been demonstrated at potential end-use, Swinkels Brewery, and is being scaled up as we speak.
In this video, the process of Iron Fuel is explained further:
SOLID has not been working on this technology alone but worked in a consortium with many partners. These are: the TU/e, Metalot, EMGroup, HeatPower, Pometon, RIFT, RI.SE, Shell, and Romico. And the projects of this technology are supported by: Uniper, Enpuls, Nyrstar, Veolia, Swinkels, RWE, and Energy Storage NL.
Our Team
SOLID is a student team of approximately 20 students every academic year. The team consists of full-time members and part-time members. We have several sub-teams in our team. Starting with the management team, making sure all subteams know what they have to do. The business team is working on the economic feasibility of IRHYS technology. The marketing team is promoting IRHYS but also Iron Fuel, making sure everybody knows the technology exists.

The technical team is designing and building the reactor. And we also have our finance team, checking all the expenses and making sure we have enough money. At last, we have our HR team, keeping everybody in the team happy! The members of our team all have different educational backgrounds. Some are still in their bachelor’s or are just finished with their bachelor’s, while others are already in their masters. We as SOLID think it is important to help with the energy transition and contribute to a greener tomorrow. We strive to enable access to clean and renewable energy for anyone at any time!
References
[1] Brinkman, L., Bulfin, B., & Steinfeld, A. (2021). Thermochemical Hydrogen Storage via the Reversible Reduction and Oxidation of Metal Oxides. Energy & Fuels, 35(22), 18756–18767. https://doi.org/10.1021/acs.energyfuels.1c02615
About the University Technology Exposure Program 2022
Wevolver, in partnership with Mouser Electronics and Ansys, is excited to announce the launch of the University Technology Exposure Program 2022. The program aims to recognize and reward innovation from engineering students and researchers across the globe. Learn more about the program here.