3D Printing in Healthcare: Engineering Applications and Impact
A technical analysis of 3D printing in healthcare, covering materials, clinical applications, and regulatory considerations for engineers.

3D printing in healthcare is now embedded in clinical practice, not just confined to prototyping labs. Surgeons plan procedures with 3D printed anatomical models, dentists fabricate patient-specific devices in-house, and pharmaceutical teams explore new ways to structure drug delivery. This shift in 3D printing in healthcare is driven by the alignment of high-resolution printing systems, biocompatible materials, and patient-specific imaging data from CT and MRI, enabling geometries and customization that conventional manufacturing cannot realistically achieve.
This article examines the engineering foundations of 3D printing in healthcare. It outlines the principal printing technologies, with attention to their resolution and throughput characteristics, and reviews the classes of materials currently qualified for clinical use. It also maps the major application domains, from surgical planning and dental devices to implants and bioprinting.
The discussion extends to the regulatory frameworks that govern medical additive manufacturing, alongside the technical constraints that still limit broader adoption. The aim is to give engineers and technical professionals a clear basis for evaluating where additive manufacturing fits within healthcare product development and clinical workflows.
Important Uses of 3D Printing in Healthcare
Healthcare has become one of the most technically mature uses of additive manufacturing. What began as a tool for rapid prototyping is now part of routine clinical workflows in surgical planning, dentistry, orthopedics, and personalized drug development. Regulators have also moved from treating medical 3D printing as an emerging niche to treating it as a defined manufacturing pathway, with the U.S. Food and Drug Administration continuing to expand oversight of printed devices, patient-matched implants, and point-of-care production inside hospitals.
The practical value of medical 3D printing lies in customization. Conventional manufacturing is efficient when every part is identical, but healthcare often requires the opposite: devices tailored to a single anatomy, a single pathology, or even a single dosage profile. Additive manufacturing makes that level of personalization economically feasible at clinical scale.
Surgical Planning and Anatomical Models
Patient-specific anatomical models generated from CT or MRI data allow surgeons to examine complex structures before entering the operating room. Cardiac surgeons, neurosurgeons, and maxillofacial teams now use printed replicas to rehearse procedures, evaluate access routes, and explain treatment options to patients. The strongest evidence continues to show reductions in operating time and improved procedural confidence in complex cases, particularly where anatomy is difficult to interpret on a screen alone.[1]
These models represent some of the simplest medical 3D printing applications, as they have no specific biocompatibility or strength requirements. They are typically produced with fused deposition modeling for rapid low-cost parts, stereolithography for fine vascular detail, or material jetting when multiple tissue types must be represented in a single model. Hospital-based printing centers have expanded steadily as institutions bring design and production closer to the point of care rather than relying entirely on outside suppliers.
Custom Implants
Custom implants are one of the most established clinical uses of additive manufacturing. Patient-specific cranial plates, mandibular reconstruction components, spinal cages, and porous acetabular cups can be designed directly from imaging data when standard implants do not provide a reliable fit. Metal powder bed fusion is the dominant production method because it can manufacture titanium structures with controlled porosity that encourage bone integration while maintaining mechanical strength.
The workflow usually combines image segmentation, CAD modeling, finite element validation, and surgeon review before manufacturing. Because the implant is built directly from digital data, the process can reduce intraoperative modification and improve alignment with surrounding anatomy. The FDA now treats these devices within a clearer regulatory framework, but process validation and traceability remain central requirements for clinical use.
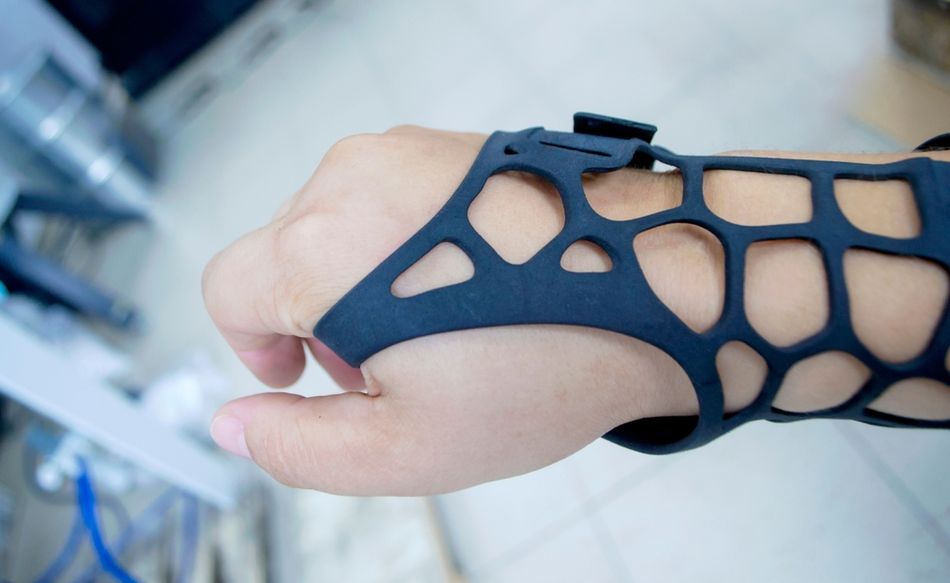
Prosthetics and Orthotics
3D printing has also changed prosthetics and orthotics by reducing cost and shortening production time. Traditional fabrication often depends on manual molding and repeated fittings, while digital workflows can capture patient geometry through scanning, adjust the design in software, and produce a device within days. For pediatric patients, who outgrow devices quickly, that speed can have a meaningful clinical advantage.
Selective laser sintering and fused filament fabrication are commonly used for braces, ankle-foot orthoses, wrist splints, and lightweight upper-limb prostheses. The ability to vary stiffness across a single part also allows engineers to design structures that improve comfort without sacrificing support.[2] Low-cost prosthetics and orthotics were one of the most visible applications of consumer-grade 3D printers during their initial wave of popularity in the 2010s.
Dental Applications
Dentistry is one of the largest commercial segments of healthcare 3D printing. Printed dental models, surgical guides, clear aligner molds, temporary crowns, dentures, and metal frameworks are now integrated into digital dental workflows. In many practices, intraoral scanning, CAD design, and resin printing have compressed production times from weeks to a single day.
The combination of high-volume demand, repeatable geometry, and relatively small part size makes dental manufacturing especially well suited to additive methods. Much of the sector’s growth has come from validated resin systems and desktop printers designed specifically for clinical environments. Align Technology, creator of Invisalign, is the world’s biggest user of 3D printing across any industry, deploying the technology to produce around a million patient-specific molds per day.[3]
Pharmaceutical and Drug Delivery
Additive manufacturing has also entered pharmaceutical development. Back in 2015, the FDA-approved epilepsy drug Spritam demonstrated that 3D printing could produce tablets with internal structures impossible to create through conventional compression.[4] Researchers are now exploring personalized dosage forms, multi-drug tablets, and implantable drug systems that release medication at controlled rates over time.
Most of this work remains in research rather than mainstream production, partly because pharmaceutical regulation is still adapting to individualized manufacturing. Even so, the technology has opened a credible path toward patient-specific medicine rather than one-size-fits-all dosing.
Bioprinting and Tissue Engineering
Bioprinting remains the most experimental branch of healthcare additive manufacturing. Instead of printing inert polymers or metals, these systems deposit living cells within hydrogel matrices to create tissue-like structures. Current work focuses on skin substitutes, cartilage repair, bone scaffolds, and miniature organ models used for drug screening.
The long-term objective is functional organ fabrication suitable for transplantation, but major engineering barriers remain. Vascularization, immune compatibility, long-term cell viability, and reproducible manufacturing still limit translation into routine clinical care. For now, bioprinting is less important as a replacement for transplantation than as a research platform for modeling human tissue more accurately than conventional laboratory methods.
Polymer 3D Printing in Healthcare
Polymers remain the dominant material class in medical additive manufacturing because they sit at the intersection of processability, cost, and regulatory familiarity. They can be engineered across a wide spectrum—from rigid, load-bearing implants to flexible, bioresorbable scaffolds—and are compatible with all major printing modalities, including extrusion, vat photopolymerization, and powder bed fusion.
Just as important, polymer workflows integrate cleanly with clinical imaging and digital design pipelines, making them well suited to point-of-care production. The tradeoff is that no single polymer meets all clinical requirements: mechanical strength, sterilization resistance, long-term biocompatibility, and degradation behavior must be balanced against printability and throughput. As a result, material selection is tightly coupled to both the application and the manufacturing process.
The current landscape can be understood as a set of overlapping material families, each aligned to specific clinical use cases and printing technologies:
PLA (polylactic acid): Biodegradable thermoplastic used for anatomical models, surgical guides, and temporary fixation concepts. It degrades via hydrolysis over time, but brittleness and low thermal resistance limit load-bearing and long-term implant use. Main advantage is low cost and ease of printing in clinical environments.
PEEK (polyether ether ketone): High-performance thermoplastic used in cranial implants, spinal cages, and orthopedic components. Its bone-like stiffness, radiolucency, and chemical resistance make it suitable for permanent implants. Requires high-temperature printing systems and tight process control.
PEKK (polyether ketone ketone): Similar to PEEK but with a wider processing window and improved printability. Used in patient-specific implants and trauma devices where mechanical stability and chemical resistance are required.
Nylon (PA 12, PA 11): Widely used in powder bed fusion for prosthetics, orthotics, and durable medical parts. Offers good fatigue resistance and balanced mechanical properties, making it suitable for wearable and functional clinical devices.
Photopolymer resins: UV-curable materials used in SLA and DLP for dental models, surgical guides, and anatomical replicas. Biocompatible grades exist for short-term contact, but brittleness limits long-term implant use.
Bioresorbable polymers (PLLA, PGA, PCL): Used in temporary implants, scaffolds, and drug delivery systems. Designed to degrade predictably in vivo, with tunable mechanical and resorption behavior, but limited in high-load applications.
Fused Deposition Modeling (FDM)
FDM/FFF, or material extrusion, deposits thermoplastic filament layer by layer through a heated nozzle. In medical applications, it is valued for its low cost, accessibility, and compatibility with a broad range of polymers. Hospitals and labs often use FDM for anatomical models, surgical guides, and low-load prosthetic components, especially in point-of-care settings.
The process produces visible layer lines and comparatively rough surface finishes. The process supports printing materials such as PLA, PETG, ABS, nylon, and high-performance polymers like PEEK, although the latter requires nozzle temperatures of around 400 °C and heated build chambers. FDM parts exhibit anisotropic mechanical properties, with weaker strength along the build axis, which limits their use in critical load-bearing applications. Despite these constraints, the speed and flexibility of FDM make it a practical tool for rapid iteration and low-cost customization.
Recommended reading: PEEK 3D Printing: Unlocking the Potential of High-Performance Polymers
Vat Photopolymerization
Vat photopolymerization processes such as SLA and DLP use light to cure liquid resin into solid parts with high precision. SLA traces each layer with a laser, while DLP projects an entire layer simultaneously, improving speed. Both methods achieve fine layer resolutions, typically in the range of 25 to 50 micrometers, and produce smooth surfaces with high geometric fidelity.
These characteristics make SLA and DLP the dominant technologies in dental manufacturing, where they are used for aligners, crowns, bridges, surgical guides, and detailed anatomical models. They are also widely used in audiology for custom hearing aid shells and in microfluidics for lab-on-chip devices. The availability of certified biocompatible resins has enabled broader clinical adoption, particularly for short-term contact applications.
Limitations include the relatively brittle nature of many cured resins, the need for post-processing (washing and UV post-curing), and a narrower range of mechanical performance compared with thermoplastics or metals. Long-term implant use remains limited.
Selective Laser Sintering (SLS)
SLS is a powder bed fusion process in which a laser selectively fuses polymer powder—most commonly nylon—within a heated chamber. Because parts are supported by the surrounding powder, complex geometries can be produced without dedicated support structures, including internal channels, lattice structures, and undercuts.
In healthcare, SLS is widely used for prosthetic sockets, orthotic devices, surgical instruments, and functional end-use parts. The resulting components have relatively uniform mechanical properties, with tensile strength typically in the 45–50 MPa range for PA 12, and better isotropy than extrusion-based methods. Surface roughness is moderate but can be improved through finishing processes such as tumbling, coating, or vapor smoothing.
SLS occupies a middle ground between low-cost extrusion and high-resolution resin printing, offering a combination of mechanical performance, geometric freedom, and scalability that makes it particularly well suited to patient-specific wearable devices and durable clinical components.
Metal 3D Printing in Healthcare
Metal additive manufacturing occupies the high-performance end of the clinical spectrum, where mechanical strength, fatigue resistance, and long-term biocompatibility are non-negotiable. Unlike polymer-based systems, which dominate models and short-term devices, metal processes are used for permanent implants that must withstand cyclic loading, integrate with bone, and remain stable in the body for decades. The key advantage is not just strength, but design freedom: lattice structures, porous interfaces, and patient-specific geometries can be engineered directly into the implant in ways that conventional machining or casting cannot achieve.
At the same time, metal AM introduces tighter process controls and higher regulatory scrutiny. Microstructure, porosity, residual stress, and surface condition all directly influence implant performance, so manufacturing workflows are closely coupled with post-processing, inspection, and validation. As a result, metal 3D printing is typically concentrated in specialized production environments rather than point-of-care settings.
The primary materials in clinical use reflect a balance between biocompatibility, mechanical behavior, and manufacturability:
Titanium (Ti-6Al-4V): The most common material for load-bearing implants in orthopedics and craniomaxillofacial surgery. It offers high strength-to-weight ratio, corrosion resistance, and strong biocompatibility. Its relatively low modulus helps reduce stress shielding, and porous or lattice structures support osseointegration. Used in spinal cages, hip components, cranial plates, and trauma implants.
Cobalt-chromium (CoCr): Used in dental frameworks, partial denture bases, and wear-resistant orthopedic parts. It has higher stiffness than titanium and excellent wear resistance, making it ideal for thin, durable structures and articulating components, though its rigidity can increase stress shielding.
Stainless steel (316L): Used mainly for surgical instruments, guides, and temporary fixation devices. It is cost-effective and easy to process, but less common for permanent implants due to corrosion concerns and nickel content.
Emerging alloys: Tantalum is used in porous implants for strong bone ingrowth, while nickel-titanium (Nitinol) enables shape-memory devices. Both are more specialized due to processing constraints but expand functional options in metal additive manufacturing.
Metal Powder Bed Fusion (PBF)
Metal powder bed fusion is the dominant manufacturing approach for medical metal AM, encompassing laser-based systems (often referred to as SLM or L-PBF) and electron beam melting (EBM). In both cases, a focused energy source fully melts metal powder particles layer by layer, producing dense, near-wrought material properties when properly processed.
In healthcare, metal PBF is used to produce patient-specific and standardized implants, including acetabular hip cups, spinal cages, cranial and maxillofacial plates, dental frameworks, and specialized surgical instruments. One of its defining capabilities is the integration of porous lattice structures directly into the implant surface. These structures, often designed to mimic trabecular bone, improve mechanical fixation and encourage bone ingrowth, reducing the need for secondary coatings or cement fixation.
The two primary variants differ in their process environments and outcomes:
Laser-based systems operate in an inert gas atmosphere (typically argon), offering finer feature resolution and better surface finish, which is advantageous for detailed geometries and thinner structures.
EBM systems operate under vacuum and at elevated build temperatures, which reduces residual stress and can improve build stability for larger titanium implants, though with coarser surface finish and lower resolution.
Post-processing is not optional—it is integral to the final part quality. Typical steps include stress relief heat treatment to reduce residual stresses, removal of support structures, machining of critical interfaces, and surface finishing through bead blasting, polishing, or chemical treatments. In many cases, hot isostatic pressing (HIP) is used to reduce internal porosity and improve fatigue performance. Final inspection involves dimensional metrology, non-destructive evaluation (such as CT scanning), and mechanical testing to ensure compliance with medical device standards.
Despite its advantages, metal PBF remains constrained by cost, throughput, and process complexity. Build rates are relatively slow, machines are capital-intensive, and powder handling requires strict controls. However, for high-value, patient-specific, or geometrically complex implants, these tradeoffs are often justified—making metal additive manufacturing one of the most clinically impactful segments of the field.
Ceramic 3D Printing in Healthcare
Ceramics occupy a distinct niche in medical additive manufacturing, defined less by structural load-bearing performance and more by biological function. Unlike metals, which are optimized for strength, or polymers, which balance flexibility and processability, ceramics are primarily used where bioactivity, wear resistance, or chemical stability are critical. In practice, this means bone regeneration, dental restorations, and specialized coatings that interact directly with patient anatomy at the interface between implant and tissue.
The clinical value of ceramics lies in their ability to mimic or integrate with natural biological structures. Calcium phosphate ceramics, for example, closely resemble the mineral phase of bone, making them inherently compatible with bone remodeling processes. At the same time, ceramic processing is more complex than polymer or even metal workflows, requiring multi-stage fabrication and high-temperature sintering, which limits true on-demand production in most healthcare settings.
Ceramic materials used in biomedical additive manufacturing generally fall into three categories:
Hydroxyapatite (HA): A bone-like calcium phosphate used as implant coatings and bone scaffolds. Supports osseointegration and bone growth but is brittle, limiting load-bearing use. Printed via binder jetting or extrusion with post-sintering.
Tricalcium phosphate (TCP): A bioresorbable scaffold material for bone grafts. Gradually degrades and is replaced by natural bone, with 3D printing enabling control over porosity and resorption rate.
Zirconia: A strong ceramic used mainly in dental crowns, bridges, and implants. Combines high strength and good aesthetics. Typically 3D printed via slurry-based processes followed by debinding and sintering.
Binder Jetting
Binder jetting deposits a liquid binding agent onto a bed of ceramic powder, selectively joining particles layer by layer. The printed part is initially fragile and porous, requiring post-processing steps including curing, debinding, and sintering. This approach is widely used for hydroxyapatite and TCP scaffolds, particularly in bone regeneration research and clinical applications.
One of the main advantages is geometric freedom. Complex porous structures can be fabricated without support structures, making it possible to design scaffolds that closely match patient anatomy and support vascularization. This is especially relevant in pre-surgical planning and reconstruction of irregular bone defects. However, shrinkage during sintering must be carefully controlled to maintain dimensional accuracy.
Recommended reading: Binder Jetting: A Comprehensive Guide to the Additive Manufacturing Process
Material Extrusion (Robocasting / Direct Ink Writing)
Extrusion-based ceramic printing uses highly loaded ceramic pastes or inks that are deposited through a nozzle. This method is commonly used for calcium phosphate scaffolds, where porosity and channel architecture are critical for tissue ingrowth and nutrient transport.
The process is relatively simple and compatible with a range of materials, but resolution is lower than photopolymer-based methods. It is often used in research and custom biomedical applications rather than high-throughput production. Its ability to produce scaffolds tailored to patient anatomy makes it relevant for healthcare providers working in regenerative medicine.
Vat Photopolymerization
In this approach, ceramic particles are suspended in a photopolymer resin, which is selectively cured using light. After printing, the part undergoes debinding to remove the polymer matrix, followed by sintering to densify the ceramic. This method is widely used for zirconia dental restorations and high-precision components.
The key advantage is resolution and surface finish, which are significantly better than extrusion or binder jetting. This makes it suitable for applications requiring tight tolerances, such as crowns, bridges, and other dental devices. The limitation is process complexity, including material handling, shrinkage control, and longer production times compared with polymer systems.
3D Bioprinting
3D bioprinting extends the use of 3D printing from inert structures into living systems, where the objective is not just to replicate form, but to recreate biological function. Instead of polymers or metals, the 3D printing process deposits bioinks—combinations of living cells and supportive biomaterials—into organized, tissue-like architectures.
These constructs are designed to mimic patient anatomy at the cellular level, making bioprinting especially relevant for biomedical research, drug development, and, longer term, regenerative medicine. For medical professionals, the value lies in improving patient care through better disease models, more predictive testing, and eventually implantable tissues.
At the core of bioprinting is the bioink. A viable bioink must balance printability, mechanical stability, and biological compatibility, ensuring that cells survive the printing process while maintaining their ability to proliferate and differentiate. Most bioinks are hydrogel-based, providing a hydrated environment similar to the natural extracellular matrix.
Alginate-based bioinks: Derived from seaweed, alginate is widely used due to its low cost and rapid gelation in the presence of calcium ions. It enables straightforward cell encapsulation and is compatible with extrusion systems. However, alginate lacks natural cell-binding sites, which can limit cell adhesion unless modified.
Gelatin methacryloyl (GelMA): A photo-crosslinkable hydrogel derived from gelatin, GelMA supports cell adhesion and proliferation while offering tunable mechanical properties. Adjusting the degree of methacrylation and light exposure allows control over stiffness and degradation behavior. It is one of the most widely used materials in bioprinting research.
Collagen and fibrin: These extracellular matrix proteins promote cell attachment and growth, making them useful in skin, vascular, and cartilage models. Their main limitation is low mechanical strength, so they are often combined with other materials for reinforcement.
Decellularized extracellular matrix (dECM): Derived from processed donor tissues, dECM bioinks retain tissue-specific proteins and growth factors. This helps guide cell differentiation in organ-specific models such as liver or cardiac tissue. Variability and limited mechanical strength remain challenges.
Bioprinting technologies differ in how they deposit bioinks, with each approach balancing resolution, cell viability, and material compatibility. The choice of method depends on the intended application, whether for high-resolution tissue modeling or larger scaffold fabrication for complex procedures.
Extrusion-based bioprinting
Extrusion is the most widely used approach in the current use of 3D printing for biological systems. It pushes bioink through a nozzle as a continuous filament, enabling high cell densities and compatibility with a broad range of materials. This makes it well suited to larger tissue constructs used in pre-surgical planning, disease modeling, and other complex procedures. The tradeoff is lower resolution and potential shear stress on cells.
Platforms from companies like CELLINK (BICO Group) and Aspect Biosystems are widely used by healthcare providers and research labs, reflecting how extrusion systems have become standard biomedical tools.[5]
Inkjet bioprinting
Inkjet systems deposit small droplets of bioink with high spatial precision, allowing fine control over cell placement. This makes them valuable for simulation, drug screening, and micro-scale tissue models where accuracy is more important than structural strength. However, they are limited to low-viscosity materials and lower cell densities.
Companies such as VivoSim Labs (Organovo) have used droplet-based approaches to develop tissue models for pharmaceutical testing, particularly in workflows that complement traditional manufacturing and testing methods.[5]
Laser-assisted bioprinting
Laser-assisted methods use focused energy to deposit bioink without a nozzle, enabling high precision and strong cell viability. This makes them suitable for advanced biomedical applications, including vascularized tissue constructs and detailed research models derived from radiology data. The downside is cost and complexity, which limit widespread adoption.
Firms like Poietis are developing these systems for specialized applications, often in collaboration with device manufacturers exploring next-generation medical equipment.[6]
Regulatory and Quality Considerations
As the use of 3D printing in healthcare expands across patient care, surgical planning, and device manufacturing, regulatory frameworks have adapted to ensure that devices produced through additive manufacturing meet the same safety and performance expectations as those made via traditional manufacturing. Regulators do not treat the 3D printing process as a separate category of medical technology; instead, it is evaluated within established medical device frameworks, with additional scrutiny on digital design control, process validation, and variability introduced across the 3D printing process.
In practice, regulatory requirements depend on how applications of 3D printing are deployed in clinical workflows. Anatomical models used for simulation and pre-surgical planning may be regulated differently from implants or instruments used directly in a surgical procedure. This distinction becomes especially important when healthcare providers operate on-site production systems for on-demand manufacturing, where responsibility may overlap with that of device manufacturers producing regulated medical equipment.
FDA Framework (United States)
The U.S. Food and Drug Administration regulates 3D printed medical devices under the same risk-based classification system applied to conventionally manufactured medical equipment. The pathway depends on device risk class, intended use, and whether an existing predicate device is available:
510(k) clearance: The most common pathway, used when a device is substantially equivalent to an already marketed product. Many 3D printed surgical guides, anatomical models used in radiology-supported diagnosis, and standard orthopedic and dental implants fall under this route. These products are widely used by medical professionals for pre-surgical planning and improving procedural accuracy in complex procedures.
De Novo classification: Used for novel, low- to moderate-risk devices without a predicate. This pathway is increasingly relevant for patient-specific devices enabled by digital design and on-demand manufacturing, particularly where customization to patient anatomy is central to function.
Premarket Approval (PMA): Required for high-risk Class III devices, including many permanent implants and load-bearing components such as certain orthopedic and cardiovascular devices. PMA requires extensive clinical data demonstrating safety and effectiveness in patient care settings.
The FDA’s foundational guidance remains Technical Considerations for Additive Manufactured Medical Devices (2017), which continues to underpin regulatory submissions.[7] It addresses process validation, build orientation effects on mechanical properties, powder and resin handling, post-processing controls, biocompatibility testing aligned with ISO 10993, and cleaning validation to remove residual materials that could affect patient outcomes.
As of the mid-2020s, the FDA has cleared or approved more than 200 devices incorporating 3D printing, spanning orthopedic implants, dental implants, surgical instruments, and patient-specific anatomical models used in simulation and surgical planning.[8]
ISO and ASTM Standards
International standards define the quality systems that support the use of 3D printing in healthcare across device manufacturers, healthcare providers, and biomedical supply chains. These frameworks ensure consistency across the full 3D printing process, from digital design through fabrication and sterilization:
ISO 13485: Quality management systems for medical devices, covering the entire lifecycle of medical equipment production, including additive manufacturing workflows.
ISO 10993: Biological evaluation of medical devices, governing biocompatibility testing for materials used in patient contact.
ISO/ASTM 52900: Standard terminology for additive manufacturing.
ISO/ASTM 52910: Design guidelines for additive manufacturing, including geometric constraints and functional requirements.
ISO/ASTM 52920: Qualification principles for additive manufacturing production sites and process validation.
These standards are increasingly important as applications of 3D printing move from prototyping into regulated production of patient-specific devices, particularly in areas involving complex procedures and long-term implantation.
Point-of-Care Manufacturing and Hospital-Based Printing
A major shift in the use of 3D printing in healthcare is the rise of point-of-care manufacturing, where hospitals and healthcare providers produce devices internally. This includes 3D printed anatomical models used for simulation, surgical guides derived from radiology data, and, in select cases, patient-specific devices used directly in a surgical procedure.
In these environments, healthcare providers may effectively assume responsibilities similar to device manufacturers, requiring formal quality systems, validation protocols, and traceability across the 3D printing process. The regulatory burden depends on intended use: anatomical models used for visualization generally face lower requirements, while devices that directly influence surgical outcomes or patient outcomes are subject to higher scrutiny.
Conclusion
3D printing now holds an established role in healthcare, supported by a growing base of cleared devices, validated clinical workflows, and an expanding range of materials. It delivers clear value in surgical planning through 3D printed anatomical models that improve patient outcomes, in patient-specific implants with improved anatomical fit, in prosthetics enabled by on-demand production and lower costs, and in dental applications where digital workflows improve efficiency for healthcare providers and device manufacturers.
Key engineering constraints still limit broader adoption. Build speed, mechanical anisotropy, material qualification, and regulatory compliance remain central challenges across the 3D printing process, particularly as applications of 3D printing move from prototypes to regulated medical equipment used in patient care. Bioprinting remains the most ambitious frontier, with functional tissue still far from routine clinical use.
For engineers, the priority is matching the right technology and range of materials to the clinical requirement—whether simulation, pre-surgical planning, or complex procedures—while ensuring a clear regulatory pathway and robust quality systems. The field is advancing quickly, and organizations that combine strong engineering foundations with clinical collaboration and radiology-driven design are best positioned to improve patient outcomes.
FAQ
What 3D printing technologies are used in healthcare?
FDM is used for anatomical models and low-cost prosthetics, SLA/DLP for dental work and high-detail models, SLS for durable orthotics and prosthetics, and metal powder bed fusion (SLM/EBM) for load-bearing implants. Extrusion-based bioprinting is mainly used in research for tissue engineering.
What materials are used in approved medical 3D printing?
Common materials include titanium (Ti-6Al-4V) and cobalt-chromium for implants, PEEK for spinal and cranial devices, biocompatible photopolymer resins for dental guides and restorations, and nylon (PA 12) for orthotic devices. Approval is device and process specific under FDA biocompatibility standards.
How does 3D printing improve surgical outcomes?
3D printed anatomical models from CT or MRI help surgeons understand patient anatomy and rehearse procedures. This can reduce operating time, lower blood loss, and improve outcomes in complex procedures such as cardiac, craniofacial, and tumor surgeries. Surgical guides also improve implant accuracy.
What is the regulatory pathway for 3D printed devices?
Devices follow standard FDA pathways: 510(k) for predicate-based devices, De Novo for novel low to moderate risk devices, and PMA for high risk implants. FDA guidance on additive manufacturing covers validation, testing, and post-processing requirements.
What limits bioprinting in clinical use?
Key challenges include creating vascular networks, maintaining cell viability, scaling to functional organs, ensuring mechanical stability, and limited regulatory frameworks for living tissues. Most current use is in research and drug testing rather than transplantation.
Can hospitals manufacture 3D printed devices on site?
Yes. Many hospitals operate point-of-care 3D printing labs to produce anatomical models and surgical guides. These facilities must follow quality system requirements, and regulatory oversight depends on whether the output is for visualization or direct surgical use.
How does 3D printing reduce prosthetics cost?
It replaces manual molding and casting with digital design and automated production, reducing labor and material costs. This enables cheaper, faster prosthetics and easier replacements, especially important in pediatric care where devices are frequently resized.
References
[1] Montgomery SJ, Kooner SS, Ludwig TE, Schneider PS. Impact of 3D printed calcaneal models on fracture understanding and confidence in orthopedic surgery residents. Journal of surgical education. 2020 Mar 1;77(2):472-8.
[2] Um HJ, Kim HS, Hong W, Kim HS, Hur P. Design of 3D printable prosthetic foot to implement nonlinear stiffness behavior of human toe joint based on finite element analysis. Scientific Reports. 2021 Oct 5;11(1):19780.
[3] Dave, P. (2026, March 17). How Invisalign became the world’s biggest 3D printing company. WIRED.
[4] U.S. Food and Drug Administration. Spritam (levetiracetam) tablets for oral suspension: NDA 207958 Approval Package. Silver Spring (MD): FDA; 2015 [cited 2026 Apr 16].
[5] Sokolova D. Five companies leading the way in 3D bioprinting. BiopharmaTrend; 2025 Nov 4 [cited 2026 Apr 16].
[6] Poietis. Laser bioprinting: from microscale precision to engineered tissue solutions. Pessac (France): Poietis; [cited 2026 Apr 16].
[7] U.S. Food and Drug Administration (FDA). Technical considerations for additive manufactured medical devices: guidance for industry and FDA staff. Silver Spring (MD): FDA; 2017 [cited 2026 Apr 16].
[8] Fortune Business Insights. 3D printing medical devices market size, share and COVID-19 impact analysis, by products, technology, application, end user and regional forecast, 2026–2034. Pune (India): Fortune Business Insights; 2026 [cited 2026 Apr 16].