NiMH vs Lithium Ion Batteries: A Comprehensive Comparison for Engineers
Choosing the optimal battery technology is pivotal to avoid future consequences. This comprehensive guide delves into the intricacies that distinguish NiMH and Lithium Ion batteries – their fundamental properties, performance across applications, etc. and equips readers for informed decision-making.
While nickel-metal hydride (NiMH) and lithium-ion (Li-ion) batteries play essential roles in engineering systems, they have different applications.
NiMH batteries replaced the older nickel-cadmium batteries and tend to be more cost-effective than lithium-ion batteries, with a life cycle of roughly two to five years [1]. They are often used in consumer electronics, hybrid vehicles, and medical devices. On the other hand, lithium-ion batteries have a high energy density and a life cycle of about five years. Lithium-ion batteries also charge faster and do better in extreme weather conditions. They are used in grid energy storage systems, electric vehicles, and portable electronics.
So, how do the batteries differ? The key differences lie in how the batteries perform. Engineers must consider safety, cost, power density, and cycle life when selecting the best battery technology for their applications. Understanding these differences can help improve efficiency and reduce safety risks.
Understanding the Basics
NiMH Battery Fundamentals
Nickel-Metal Hydride (NiMH) batteries consist of a positive cathode (nickel hydroxide) and a negative anode (a hydrogen-absorbing alloy). Each NiMH battery cell has a voltage of 1.25V.
The Charging Process
During the charging process, the positive cathode or nickel hydroxide undergoes oxidation, releasing electrons. And the negative anode is decomposed into Hydrogen atoms.
The reaction [2] is as follows:
At the positive electrode, the reaction is:
Ni(OH)2 + OH- becomes NiOOH + H2 O + e-
At the negative electrode, the reaction is:
Alloy + H2 O + e- becomes Alloy (H) + OH-
The Discharge Process
At the negative electrode, the hydrogen is desorbed and reacts with a hydroxyl ion to form water:
At the negative electrode, the reaction is:
Alloy (H) + OH- becomes Alloy + H2 O + e-
At the positive electrode, the reaction is:
NiOOH + H2 O + e- becomes Ni(OH)2 + OH-
This flow of electrons through the external circuit is what produces electrical energy. The charging/discharging process means NiMH batteries can be recharged hundreds of times.
It's important to note that the NiMH battery is designed to be positive-limited. This means that the negative electrode always carries a greater capacity than the positive electrode.
Suggested Readings: Students Make a Battery That Can Be Wirelessly Recharged up to 500 Times
The Key Components of a NiMH Battery Cell
A NiMH battery cell comprises a positive electrode, a negative electrode or anode, a separator, and an electrolyte.
- Positive Electrode (Cathode): The positive electrode of a NiMH battery is made of nickel hydroxide (Ni(OH)2) and an active material that can differ. The properties of nickel hydroxide include a high energy density and stability. This stability is beneficial during the charge/discharge cycles.
- Negative Electrode (Anode): The negative electrode of a NiMH battery is made up of an active material, such as lanthanum, and transition metals, such as nickel. The negative electrode is coated with a conductive substrate, like nickel. This helps transport the electrons back and forth between the positive and negative electrodes.
- Electrolyte: The electrolyte in a NiMH battery is the medium for ion transport in the battery. It is alkaline in a 20% to 40% weight alkaline hydroxide solution. The pH level and concentration of the alkaline solution are key to ensuring excellent battery performance.
- Separator: The separator in a NiMH battery cell is typically non-woven polyolefin. This porous membrane separates the positive and negative electrodes and prevents electrical short circuits while simultaneously allowing ions to flow back and forth. [2]
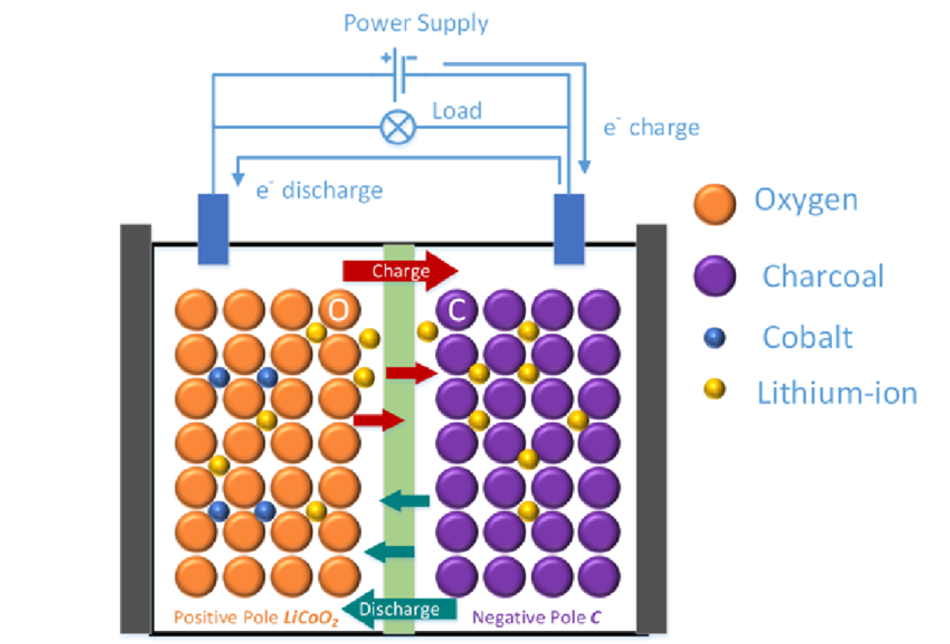
Lithium-Ion Battery Essentials
Lithium-ion batteries work by allowing lithium ions (Li+) to transfer back and forth between the positive and negative electrodes. Charging lithium-ion batteries is voltage-based as opposed to current-based.
The Charging Process
Lithium ions extract themselves from the cathode granite end and release electrons in the process. At the negative electrode end, the lithium ions are intercalated into the graphite structure, forming what is called a "lithium-intercalation" compound. The battery charges by transferring lithium ions from the positive electrode to the negative electrode.
The Discharge Process
Lithium ions at the negative electrode are ionized and are then separated from their electrons and the graphite anode. The ions return to the electrolyte solution. This reaction reduces the lithium-intercalation compounds in the negative anode. The released electrons flowing through the electrolyte solution produce electrical energy.
Why are the intercalation compounds in the electrodes crucial? The compounds allow for the lithium-ions to insert themselves into the electrodes and extract themselves from the electrodes without significant structural changes. This insertion/extraction process can take place thousands of times without any change to the battery's mechanical structure.
The Key Components of a Lithium-Ion Battery Cell
Like the NiMH battery, the Lithium-ion battery cell is made up of four main components: the cathode, anode, electrolyte, and separator.
- The Cathode: The cathode in the lithium-ion battery is the positive electrode. This is where the lithium is stored. The higher the amount of lithium, the higher the capacity of the battery. The cathode is coated with an active material to help the transfer of lithium ions. This active material is lithium oxide.
- The Anode: The anode is the negative electrode and it is made up of graphite. Like the cathode, it is also coated with an active material. When the battery is charging, lithium ions are stored in the anode.
- The Electrolyte: The electrolyte in the lithium-ion battery allows the lithium ions to move back and forth between the cathode and the anode during the charge/discharge cycles. It is made up of salts, solvents, and additives. The speed of the lithium ions depends on the electrolyte used.
- The Separator: The separator keeps the cathode and anode apart and ensures the battery is safe to use. It is a porous material that only allows lithium ions to pass through. Some separators used in lithium-ion batteries include polyethylene (PE) and polypropylene (PP).
The Advancements in Battery Technology
NiMH Battery Innovations
Recent innovations and advancements have made NiMH batteries more efficient, especially in their cycle life and energy density. Here are a few innovations that have taken place:
Energy Density
[3] The energy density is the amount of energy stored in a given volume of the NiMH battery. Researchers have experimented with the chemical composition of nickel, metal hydride alloys, and additives to boost the battery's energy. Specific changes have been upgraded to high-density negative and more efficient positive electrodes. This energy density has increased the battery's power from 200 to 1200 W kg−1 commercially. NiMH batteries can now pack more energy per unit volume.
Cycle Life
The cycle life has improved as researchers focused on improving the Ti-Ni alloy structure and composition. NiMH batteries can hold up to 85% of their charge after a year [4].
Electrode Materials and Surface Area
While researchers experiment with electrode material to develop new alloy compositions, others are focused on increasing the surface area of electrodes to increase the storage area and release of hydrogen ions.
Electrolyte Compositions
Electrolyte compositions have also been upgraded. Changing the electrolyte composition can increase the speed at which the hydrogen ions move through the electrolyte.
Cell Design
Luminous, a Swedish startup, has made significant progress with reusable nickel metal hydride (NiMH) batteries that use energy from renewable sources [6].
Examples of Advancements
Some examples showcasing these NiMH battery advancements include:
Panasonic Eneloop Batteries
- Panasonic's Eneloop rechargeable NiMH batteries let you charge them over 2,000 times, reducing waste and saving money.
- Due to the improved metal-hydride alloy lattice inside the battery, the battery is still 70% effective after 10 years.
- The voltage from the Eneloop battery ensures the voltage flows out steadily for longer. • A digital camera using an Eneloop battery can take about 4.4 times as many shots as a digital camera using a regular alkaline battery.
- Whereas standard NiMH batteries will react slower in extremely cold weather, the Eneloop batteries can be used between -20 and 50 degrees Celsius.
Sanyo HR-3UTGA Batteries
Sanyo's HR-3UTGA batteries stand out due to advancements in their electrode nanostructuring.
- The electrodes' surface area has been increased to improve the transfer of ions.
- Improved electrolyte material and composition provide faster charging rates and improved cycle life
- Electrode nanostructuring also results in higher energy density.
Duracell Rechargeable NiMH Batteries
Due to the advancements in electrode manufacturing techniques, various improvements have been made to Duracell's Rechargeable NiMH batteries:
- Long-Life Ion Core technology guarantees the batteries can be charged up to 400 times without losing quality.
- Power Preserve Technology ensures the batteries can hold their charge for up to one year when unused.
- High-density core technology ensures batteries can power devices for longer.
Toyota Prius Hybrid Vehicle
Due to the advancements in the NiMH battery's power, Toyota Prius hybrid vehicles use a NiMH battery pack as part of their system due to its high energy density, cost efficiency, and better performance under extreme temperatures.
Due to higher-energy batteries, NiMH batteries can hold more energy per unit of volume, making it a good choice for Toyota's hybrid vehicles. They can also hold a majority of their charge without self-discharging.
Lithium-ion Battery Breakthroughs
Like the NiMH batteries, lithium-ion batteries have seen several important breakthroughs.
Increased Safety
One of the disadvantages of lithium-ion batteries is that they can overheat if overcharged or exposed to high temperatures. This overheating increases the risk of fires. However, ceramic coatings and additives have improved the stability of the batteries. Moreover, using non-flammable electrolytes has also helped with thermal stability.
Electrolyte Materials
A significant breakthrough for lithium-ion batteries was replacing the liquid electrolyte with a solid state [5]. Solid-state lithium-ion batteries help increase the energy density while speeding up charging times.
Improved cell designs, like 3D [7] electrode architecture, have also resulted in more efficient charging.
Suggested Readings: Solid-State vs. Li-ion: Which Battery Tech is better for Electric Vehicles?
Cell Designs
Electrode Materials
One of the major research areas has been introducing nanomaterials, such as silicon nanoparticles, into the electrodes. This new material adds to the stability of the battery and increases its cycle life.
Electrolyte Compositions and Cycle Life
Increasing the cycle life is crucial for applications that need frequent charge/discharge cycles, such as electric vehicles. Innovations in electrolyte material and electrode coatings have helped prevent battery degradation while extending the cycle life.
Temperature Tolerance
Thermal management is another crucial area that has seen improvements. Better cell designs, such as thermal-resistant casing materials, have increased the battery's tolerance to low and high temperatures.
Examples of Lithium-Ion Advancements
Some examples showcasing these lithium-ion battery advancements include:
Tesla Electric Vehicles
Tesla's electric vehicles use 2170-type cylindrical cell lithium-ion batteries in its Model 3 electric vehicles and 4680-type cylindrical cell lithium-ion batteries in its Model Y electric vehicles. Both have enhanced energy density and longer cycle lives.
- High-capacity cathode materials are used in combination with silicon-based anodes. The cathode types used in Tesla electric vehicles include nickel cobalt aluminum (NCA), nickel cobalt manganese (NCM), and lithium iron phosphate (LFP). The first two are high-energy density while LFP is a lesser-energy density type of battery.
- Tesla's Supercharger technology provides impressive acceleration and long-range driving capabilities due to improvements to the lithium-ion battery's electrode materials.
- The electric vehicle can drive in extreme temperatures due to the additives, such as liquid cooling, to the electrolyte material.
Samsung Galaxy Note 20 Ultra Smartphone
The new smartphones are giving way to longer battery lives and faster charging speeds due to improvements made to lithium-ion batteries.
- Longer life cycle with battery life lasting 1.5 days
- High-capacity cathode materials, such as lithium nickel manganese cobalt oxide (NMC), give faster charging speeds
- Qualcomm Quick Charge and USB Power Delivery allow users to recharge their smartphones quickly. The Quick Charge feature lets users charge their smartphones from 0-50% in just 5 minutes.
- Improved thermal capabilities prevent the phone from overheating.
Panasonic 21700 Battery Cells
The 21700 battery cell for Tesla's electric vehicles is the highest-density lithium-ion battery as of 2024. Its energy density is 300Wh/ksg, which gives the vehicle an incredible amount of power.
- High-capacity cathode materials, like nickel cobalt aluminum oxide (NCA), allow for longer cycles, a necessary feature for electric vehicles.
- Updated electrode coatings and cell designs ensure better temperature tolerance.
Engineering Applications
NiMH Batteries in Action
Some real-world engineering projects that use NiMH batteries include:
Hybrid Electric Vehicles (HEVs)
Hybrid electric vehicles combine NiMH batteries with combustion engines and electric motors. This combination works to reduce emissions and reliance on gas. The NiMH batteries are designed to be energy storage units. They capture energy when drivers brake or decelerate and supply energy when they accelerate. NiMH battery packs are integrated into the vehicle's powertrain system.
Although NiMH batteries tend to have lower energy density compared to lithium-ion batteries, they can still pack enough energy to power a vehicle. These batteries are also more affordable than lithium-ion batteries, making them a cost-effective option when hybrid vehicles are mass-produced. Finally, NiMH batteries don't overheat in the way lithium-ion batteries do. This can help reduce the risk of a fire while driving.
Portable Electronics
Despite lithium-ion's dominance in the battery market, NiMH batteries are still used in specific applications. This includes digital cameras, handheld tools, and flashlights.
NiMH batteries in portable electronics are often rechargeable, and users can either recharge them, replace them, or swap them out. Typically, portable devices come with charging circuits to prevent overcharging.
NiMH batteries are ideal for devices and tools that must be charged frequently, such as digital cameras and handheld tools. They are also reliable in extreme temperatures. In addition, their affordability makes them a preferred choice for many electronics.
Renewable Energy Storage
Surprisingly, NiMH batteries are also used in smaller-scale off-grid solar power installations, such as stand-alone wind turbines. The batteries store excess energy for later use.
NiMH batteries are integrated with charge controllers (to keep the batteries from overcharging) and inverters to help manage energy flow. Sometimes, NiMH batteries are combined with supercapacitors and lead-acid batteries to create a hybrid and more reliable energy storage solution.
NiMH batteries are used for renewable energy storage because of their reliability and longevity. As mentioned, NiMH batteries can also be used in a hybrid energy storage system with other types of batteries. This can help reduce the need for expensive upgrades.
Lithium-Ion Batteries Powering the Future
Here are some engineering projects that rely on lithium-ion batteries:
Electric Vehicles
Although NiMH batteries are used in hybrid vehicles and one of Tesla's electric vehicles, lithium-ion batteries dominate the market. Lithium-ion's high energy density and long cycle lives make them ideal for the electric vehicle market.
Lithium-ion battery packs are integrated into the underbody of the electric vehicle. Due to their large size, placing them here helps distribute the vehicle's weight evenly. The vehicles also use advanced battery management systems (BMS) to monitor each cell's charging/discharging cycle.
Suggested Readings: What is an Autonomous Vehicle: A Comprehensive Guide to its Engineering Principles and Applications
Grid Storage Systems
Lithium-ion batteries pack a lot of power, which makes them ideal for storing excess energy generated by renewable sources, such as wind and solar power. Grid-scale energy storage can help stabilize electrical grids, especially during winter storms or heat waves when demand exceeds supply.
Lithium-ion battery banks are stored in large containers or buildings. The storage area is then placed near the renewable energy generation facility. The battery banks are connected to the electrical grid through specialized equipment, which allows the battery banks to charge or discharge electricity when needed.
Aerospace Applications
Lithium-ion batteries also power unmanned aerial vehicles (UAVs) or drones, electric aircraft propulsion systems, and satellites. The large energy storage capacity of lithium-ion batteries is ideal for these kinds of applications. Lithium-ion batteries are typically integrated into the aircraft or spacecraft's electrical power system.
The major reasons lithium-ion batteries dominate the market are:
- High Energy Density: Lithium-ion batteries can store a large amount of energy per unit volume or weight.
- Lightweight: Compared to the amount of energy they can pack, lithium-ion batteries are relatively lightweight, making them suitable to integrate into vehicles and unmanned aerial vehicles.
- Fast Charging Speeds: Users can charge lithium-ion batteries quickly, allowing for fewer downtimes when driving or using aerospace machinery.
- Efficient: There is minimal energy loss during the charge/discharge cycle. This high efficiency means better energy storage and distribution.
- Long Cycle Life: Lithium-ion batteries have long cycle lives, and they can go through many charge/discharge cycles without a significant loss in performance or capacity. This also translates to fewer battery replacements.
- Scalability: Lithium-ion batteries can be scaled down to fit into consumer electronics and scaled up for grid storage systems and aerospace applications.
A Comparative Analysis
Performance Metrics: NiMH vs Lithium Ion
Here are some key performance metrics between NiMH and lithium-ion batteries:
| Factors | NiMH | Li-ion |
| Specific Energy | 55-110 Wh/kg | 100-300 Wh/kg |
| Power Density | 100 - 500 W/kg | 500 - 5000 W/kg |
| Charge/Discharge Rates | Moderate. May degrade at higher rates | Higher charge/discharge rates |
| Temperature Performance | Wide operating range. -20°C to 60°C (-4°F to 140°F) | Narrow operating range. 0°C to 45°C (32°F to 113°F) |
Comparison Table
Here is a comparison table on the batteries' different features.
| Factors | NiMH | Li-ion |
| Rated Voltage | 1.25V | 2.4-3.8V |
| Self-Discharge Rate | Retains 50-80% after a year | Retains 90% after 15 years |
| Low Temperature | 20°C (-4°F) | 0°C (32°F) |
| Maximum Discharge | 15°C | 80C |
| Self-Discharge Rates (per month) | 15-30% | 2-3% |
| Cycle Life | 500 - 1000 | > 2000 |
| Battery Weight | Heavier than Li-ion | Lighter than NiMH |
| Memory Effect | Yes | No |
| Environmental Issues | Recyclable | Recyclable & limited |
Recommended Readings: What is a Flow Battery: A Comprehensive Guide to Understanding and Implementing Flow Batteries
Recommended Readings: Flow Cell Technology: A Comprehensive Guide
Cost Considerations
Cost of Raw Materials
The raw materials used to make NiMH batteries include nickel and metal hydride alloys. Because these materials are relatively abundant, NiMH batteries are somewhat inexpensive compared to lithium-ion batteries.
Lithium-ion batteries, on the other hand, rely on lithium and cobalt. Both lithium and cobalt are rare elements, and the cost of mining them drives up the price of lithium-ion batteries.
Manufacturing Cost
NiMH batteries are manufactured in four steps: preparing the electrodes, assembling the cells, filling the battery with electrolytes, and sealing the battery [8].
Lithium-ion batteries require similar steps, but quality control needs to be higher due to the raw materials used. This means manufacturing lithium-ion batteries requires more sophisticated and specialized equipment.
Assembly Cost
The assembly cost tends to vary depending on the type of NiMH battery. However, in general, the assembly cost is standard.
Lithium-ion batteries require more safety measures in the assembly stage due to the safety hazards associated with lithium. These additional safety measures drive up the price of lithium-ion batteries.
Economies of Scale
Lithium-ion batteries are being adopted into various applications at a far greater scale than NiMH batteries. This could ultimately drop the price of lithium-ion batteries as companies begin to improve their manufacturing processes (additive or 3D printing of Li-ion batteries for example).
Environmental Impact
Resource Extraction
Extracting and processing nickel to produce NiMH batteries can adversely affect the environment as it may contaminate water and create greenhouse gas emissions.
Extracting and mining lithium and cobalt to produce lithium-ion batteries may also have negative environmental consequences, such as soil degradation, habitat destruction, and water pollution.
Production
The production of NiMH batteries involves refining the metals, manufacturing the electrodes, and putting the cells together. These processes can cause greenhouse gas emissions and create waste disposal issues.
Similarly, lithium-ion batteries may damage the environment by producing electrodes and cobalt and lithium-based chemistries.
End-of-Life Management
NiMH batteries can be recycled. However, there are challenges in separating metals like nickel from cells. Improper disposal can cause environmental pollution.
Overall, it is easier to recycle lithium-ion batteries. However, with their growing demand, handling the volume of used lithium-ion batteries may become more challenging. Improper disposal can result not only in environmental pollution but also create fire hazards.
Recycling Processes
There have been improvements in recovering nickel and other metals from used NiMH batteries. These methods involve techniques like hydrometallurgical and pyrometallurgical processes, allowing companies to extract and refine the metals to be used again.
Recycling processes [9] for lithium-ion batteries have received much attention due to the growing demand for these batteries [10]. Hydrometallurgical and pyrometallurgical processes can also extract lithium, cobalt, and other metals from used batteries in the hopes of reusing them for future lithium-ion batteries. In so doing, a closed-loop system can be established to avoid mining new materials.
Materials Substitution
There is also research to find alternative metals to nickel and rare earth metals. Researchers are looking for more environmentally friendly electrode materials and electrolytes to improve sustainability.
A major concern for lithium-ion batteries is the heavy reliance on cobalt. As of 2024, EV batteries contain 20 kg of cobalt in each 100 kilowatt-hour (kWh) pack, with cobalt making up 20% of the cathode weight [11]. Researchers are looking into alternative cathode materials that can either lower the amounts of cobalt used or eliminate its use altogether. Solid-state electrolytes are being developed as well to reduce reliance on fossil materials.
Circular Economy Initiatives
Both the NiMH and lithium-ion battery industries are starting to embrace circular economy principles.
- By extracting the materials from used batteries, the industry can reuse these materials for new batteries, creating a closed-loop model.
- By ensuring proper disposal of used batteries, industries can reduce environmental pollution.
- By finding alternative and more environmentally friendly materials for electrolytes and electrode materials, industries can create more sustainable batteries.
Challenges and Limitations
NiMH Battery Drawbacks
While NiMH batteries offer many advantages, there are also some limitations. Two obvious limitations include the memory effect and self-discharge.
Memory Effect or Voltage Depression
The memory effect [12] in NiMH batteries occurs when they seem to "remember" their previous charge, resulting in reduced performance over time. This effect seems to happen when the batteries are frequently charged and discharged partially (and not entirely).
Why does the memory effect happen? The memory effect seems to occur due to the formation of crystalline structures. These structures form on the surface of the battery electrodes after partially charging and discharging the battery. Over time, the crystalline structures can obstruct the flow of ions, resulting in reduced performance.
When batteries are partially charged, specific areas of the electrodes become more active than others. This results in an uneven distribution of electrolyte and the active material. This uneven distribution eventually leads to crystalline structures forming.
Self-Discharge
Another limitation faced by NiMH batteries is their self-discharge tendencies. Self-discharge occurs when there is a slow loss of charge when the battery is not being used.
Self-discharge happens due to reactions that take place within the battery. These reactions involve electrode corrosion and electrolyte decomposition. In addition, even when a NiMH battery is not in use, ions can slowly migrate back and forth through the electrolyte. This movement results in a slow loss of charge over time. It is important to note that self-discharge happens faster at warmer temperatures. Keep your NiMH batteries at cooler temperatures to slow down the self-discharge process.
Unfortunately, these drawbacks can impact engineering applications. Here are some examples of how they may affect battery performance and system design.
In cases where reliable battery performance is critical - medical devices, for instance - the memory effect can result in reduced performance and safety concerns for any patient using NiMH batteries.
Although the memory effect can be fixed by performing a full charge and discharge cycle, this may interrupt crucial operations, such as in the use of a patient monitor pump. It may not always be practical as well.
To prevent this kind of unreliable performance, engineers may need to perform frequent battery maintenance.
Some NiMH batteries may be in standby mode or used just for emergency backup systems. This can be problematic if the battery pack has self-discharged in a crisis. This self-discharge issue can also affect video monitoring systems operating on NiMH batteries. If the batteries self-discharge, there will be gaps in the footage captured.
To combat this issue, engineers can factor in the self-discharge times and ensure that batteries are always ready to go even during long standby periods. Fortunately, the self-discharge time can be extended as advancements continue to be made with NiMH batteries. For example, the Eneloop low self-discharge battery can keep 85% of its charge even after one year on the shelf.
Lithium-Ion Battery Hurdles
Like NiMH batteries, lithium-ion batteries also have some challenges. Here are some hurdles these batteries must overcome.
Safety Concerns
Thermal runaway [13] is one of the major safety concerns with lithium-ion batteries. This happens when the batteries overheat due to overcharging or external heat exposure. This overheating can result in a fire or even an explosion.
Thermal runaway is typically due to hot spots within the battery that tend to overheat. Engineers can combat this issue by coating the batteries with flame-retardant materials or adding a type of coolant to the battery.
Limited Raw Material Availability
Another hurdle lithium-ion batteries must overcome is the issue of limited raw material availability. Lithium-ion batteries rely on lithium, nickel, and cobalt. However, cobalt is costly and there are concerns over mining it to meet the demands of electric vehicle car batteries.
How can engineers overcome these hurdles? A change in battery chemistry is one interesting development. Solid-state lithium-ion batteries could be a promising alternative to liquid-state lithium-ion batteries.
Researchers are also looking into raw materials besides lithium, such as sodium-ion batteries. This could significantly reduce the reliance on raw materials like lithium and cobalt.
There have also been advancements in materials science. Some materials in discussion are sulfur-based cathodes and silicon-based anodes. Making this switch could improve the energy density of the batteries.
Another key area of development is the manufacturing process. Different ways of manufacturing lithium-ion batteries could reduce costs. Some manufacturing techniques explored include additive manufacturing (3D printing), which could reduce manufacturing costs by 30%.
Conclusion
While NiMH and Lithium-ion batteries have a similar design, they have key differences. NiMH batteries offer lower energy and power compared to lithium-ion batteries, and they have higher self-discharge rates. They also suffer from the memory effect.
On the other hand, lithium-ion batteries offer more power density, higher energy density, lower self-discharge rates, faster charging capabilities, and longer cycles. However, lithium-ion batteries are more sensitive to temperatures compared to NiMH batteries, tend to be pricier than NiMH batteries, and can overheat considerably if overcharged.
There have been technical advancements for both NiMH and lithium-ion batteries. On the NiMH end, there have been significant improvements to the self-discharge and memory effect issue;. In contrast, on the Lithium-ion end, additives have been added to help with thermal management and safety concerns.
Lithium-ion batteries currently dominate the market given their wide-scale use in electric vehicles. Still, NiMH batteries are more affordable and may be preferable in specific engineering applications, such as small portable devices.
Ultimately, the choice between NiMH and lithium-ion batteries will depend on the specific engineering application.
Frequently Asked Questions
1. What are the major differences between NiMH vs lithium ion batteries in terms of performance?
NiMH batteries tend to provide lower energy density and power density compared to lithium-ion batteries. In addition, lithium-ion batteries tend to self-discharge at a slower rate and have a longer cycle life.
2. What are the main safety concerns associated with lithium-ion batteries?
Lithium-ion batteries tend to heat up quickly if overcharged or exposed to high temperatures. This risk - known as thermal runaway - can lead to fires.
3. How does the memory effect affect the performance of NiMH batteries?
The memory effect, or voltage depression, can reduce the capacity of NiMH batteries. Voltage depression happens when batteries are continuously charged and discharged partially (as opposed to fully). Partially charging and discharging the battery creates crystalline structures in the battery, decreasing its capacity.
4. How do temperature extremes affect the performance of NiMH vs lithium ion batteries?
Because a chemical reaction takes place within both types of batteries, temperature extremes can slow down the reaction, thus decreasing the battery capacity. High temperatures, on the other hand, can result in internal damage and overheating (thermal runaway) in lithium-ion batteries.
5. How can engineers increase safety when using lithium-ion batteries in engineering applications?
Engineers can increase safety with lithium-ion batteries by adding additives to the electrolyte material to prevent overheating. Electric vehicles add liquid coolant for example. They can also use flame-retardant materials when designing batteries.
References
[1] Batterydepot. NiMH vs old NiCa batteries. Link
[2] Energizer. NiMH batteries. Link
[3] Dragonflyenergy. Why Does Energy Density Matter In Batteries?. Link
[4] Wikipedia. NiMH batteries can retain 85% of their charge after a year. Link
[5] Wevolver. Solid-state battery. Link
[6] Chalmers. Reusable nickel metal hydride (NiMH) batteries. Link
[7] Bibliothek. Li-ion 3D architecture. Link
[8] Google Patents. NiMH Manufacturing process. Link
[9] Greensutra. Detailed facts on battery recycling. Link
[10] Weforum. Lithium shortage. Link
[11] Energy.gov. Li-ion battery’s reliance on cobalt. Link
[12] Large. Memory effect in NiMH. Link
[13] Saurenergy. Thermal runaways in Li ion batteries. Link