4 Considerations Often Overlooked When Designing Medical Devices
Here we discuss 4 considerations often overlooked when designing the medical devices to optimize your medical device and ensure the development process smoothly and rapidly.
Photo by Piron Guillaume on Unsplash
Developing medical devices is a complex and challenging process. Bringing the idea through to the initial prototype and then trial production to final commercialization, you must comply with strict regulations and safety standards. However, this is not enough. Launching a successful medical device without lengthy and costly delays and revisions relies on complying with layers of regulations, and the grasp of procedures and details during product development. Here we discuss 4 considerations often overlooked when designing the medical devices to optimize your medical device and ensure the development process smoothly and rapidly.
1. How to protect the (sensitive) components?
Overmolding is a process that creates a seamless combination with two or more materials. Typically, the overmolded material, usually thermoplastics or elastomers, covers the substrate, forming the medical devices with ergonomic and sturdy exteriors. There are several benefits of overmolding medical devices, including protecting the substrate from abrasion and corrosion and improving user experience with flexible and sanitary exteriors. But they are more than these. The sensitive components in overmolded medical devices could be time-efficient in production, well protected during usage, and prevent intellectual property theft.
2. Surface treatment should be considered along with color selection?
The same color might look different in various of surface treatment. A wide range of finishing options are available in medical devices to create different appearance and applications. Practically, surface treatment and color perception are inseparable, which means that surface treatment should be considered along with color selection. In development stage, you could ask the manufacturing supplier to produce the same-color parts in different surface treatment processes, including anodizing, plating, transparency, translucency. Those parts directly reflect the differences in same color, contributing to choosing the most optimal one.
In addition, the degree of surface finish greatly affects the way of color perception. You can effectively use a single color by treating components with matte finishes and glossy surfaces to reflect the different surface finishes. One thing should be noticed that it’s easy to see the fingerprints and smudges on the shiny mirror surface of medical equipment, but the over-rough texture provides hidden area for contaminants. The balance point should be found.
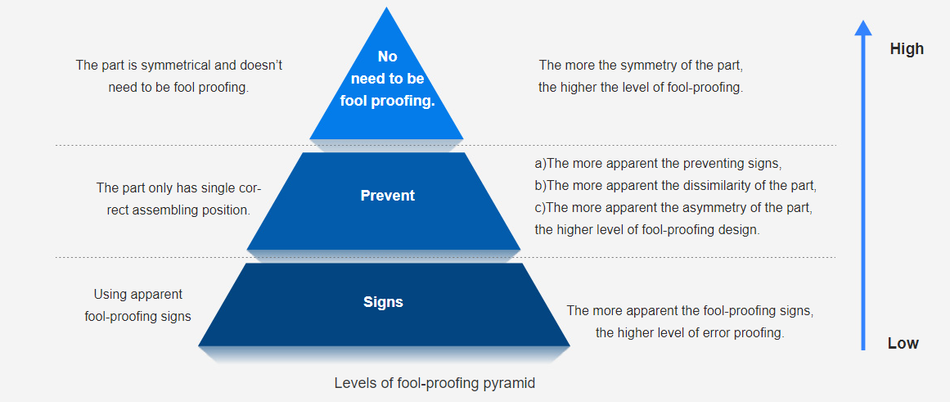
3. Are your medical parts fool-proofing?
Murphy's Law states: If anything can go wrong, it will. This is applicable to medical device.
If there are possibilities of incorrect assembly on product design, the wrong assembly is likely occurred, causing the decreased efficiency, and raised cost and time. In a similar way, if there are possibilities of misuse of medical products, then the end users may use the product in an incorrect way, leading to various consequences and even threatening life and health. Therefore, fool-proofing design is vital, which directly reduces mistakes and eliminates rework during assembly, optimizes user experience, and avoids the unnecessary cost. As Levels of Fool-Proofing Pyramid figure shows, the pyramid rises, so does the level of fool proof.
4. Do not forget the nameplates.
The nameplates of medical devices in intended environment should be tested for durability and tough enough to pass through the frequent and invasive sanitary procedures. Several materials commonly used in the medical industry include metalized polyester, polycarbonate, anodized aluminum and stainless steel, etc. Both polyester and polycarbonate nameplates are suitable for external and internal medical equipment. Metalized polyester offers shining appearance, while polycarbonate is flameresistant. Anodized aluminum and stainless steel are the common materials for nameplates to medical equipment and undergo the regular cleaning and sterilization treatments. Especially anodized aluminum offers superior resistance to abrasion and chemicals, meeting the requirements of medical equipment in sterilizing and cleaning environment.
Want to Know More?
The above are a few plain examples that might overlook in medical devices. RPWORLD offers expert technical advice and design support for customers’ medical device development.