Siemens Healthineers: Bringing point-of-care testing closer to the patient
Imagine going to the hospital with symptoms of a heart attack, only to wait an hour for the test results and treatment. With each minute ticking by, your health is in jeopardy. Siemens Healthineers is now rewriting what is possible and has set out to change this scenario in emergency departments across the world.
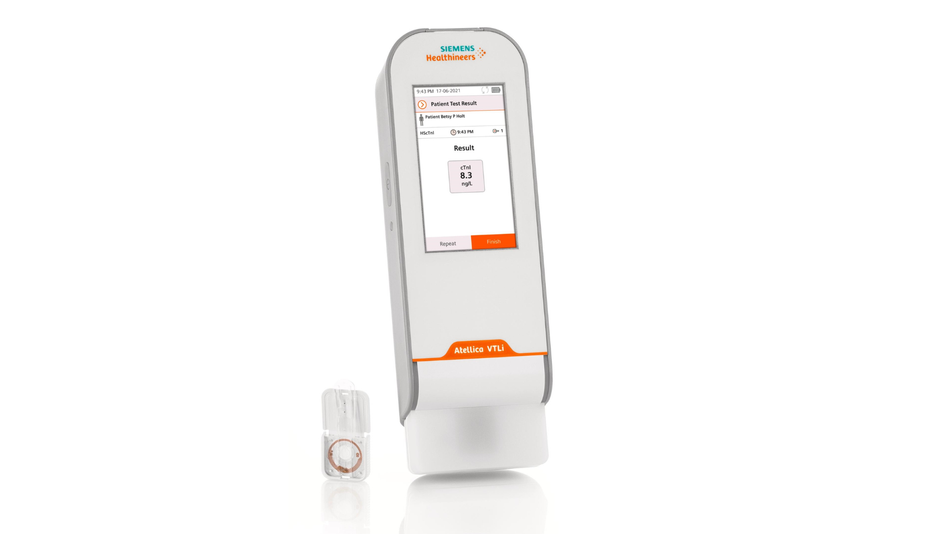
The company is bringing to market a diagnostic test for acute myocardial infarction (AMI), more commonly known as a heart attack. With its new handheld analysis device and disposable inserts, emergency departments can test a patient’s blood to diagnose a heart attack. This can be done within 10 minutes using only a single drop of blood and without ever leaving the patient’s side.
Expedited results = huge advantages
The heart attack detection now involves drawing a tube of blood, which is sent to the hospital lab for analysis and results sent back to the ED physician. This turn-around time takes about an hour. In the 50 precious minutes gained by Siemen Healthineers’ point-of-care test, doctors can immediately start treating the patient.
Expedited test results have huge advantages for the patient, the doctor and the hospital. Patients receive an early diagnosis leading to faster care and less stress during the waiting period. Doctors can quickly determine the best course of treatment for a patient. And of course, it saves the hospital money by moving patients more quickly to the correct department.
The Siemens Healthineers solution is a handheld device is called Atellica® VTLi Patient-side Immunoassay Analyzer. While it is easy to operate, the microfluidics technology inside is cutting-edge. Filtered plasma from the drop of blood goes into reaction chambers with magnetic particles which capture the troponin. These particles are manipulated and the device quantifies the amount of troponin.
In the future, this device could provide testing at other decentralized points of care.
“We are thinking further down the road. You will be able to use it as a general practitioner for all types of tests. You can also use it in the ambulance so the patient goes directly to the right hospital,” says Marcel Jacobs.
“The optical magnetic unit, as we call it, is very complicated,” says Jacobs. “The lenses that we use are on the edge of what is possible from a technological point of view.”
But although it is homegrown Eindhoven technology, it wasn’t actually Siemens Healthineers that first developed it. It was Philips.
A former Philips executive, Jacobs has been with this technology since its infancy. A medical microbiologist by training, he is now Site Head and Program Manager for Siemens Healthineers on High Tech Campus Eindhoven. He explained the biology behind the company’s solution.
Cardiac troponin is a protein released into circulation after cardiac injury and is the standard biomarker for patients presenting to the emergency department with a suspicion of AMI.
“Troponin is a marker in the blood for a heart attack. Every person has a very low level of troponin in the blood, but if you have a heart attack, then your injured myocardiac cells degrade and troponin is released. The moment you have acute heart failure, your troponin level will increase.”
Startup success story
Eight years after starting development, Philips decided to end the Atellica VTLi project (called Minicare) because the product didn't fit their portfolio.
“My colleagues and I felt the unmet need for our solution. We managed to take over all relevant assets from Philips and continue with it. We did a management buyout and founded our own company with a private investor,” Jacobs said.
The team of eight spun out the technology to form Minicare BV in 2018. They created a second-generation production and developed the high-sensitivity troponin test to the point of feasibility.
But it wasn’t a private startup for long, acquired by Siemens in 2019.
“We knew we couldn’t sell this product with such a small group. You need distribution channels and coverage in many countries,” Jacobs said. “We were looking for a commercial partner, a joint venture or an acquisition. It didn't matter to us.
Jacobs’ colleague, Jeroen Nieuwenhuis, went to the biggest diagnostic conference and approached different companies. There were several companies interested, but in the end, they had the best feeling with Siemens Healthineers.
Although the solution is now part of the larger Point-of-Care Testing business in the U.S., the technological development continues locally at Siemens Healthineers’ R&D site on HTCE and the team has now grown to 40 people.
Launching in Europe, scaling internationally
The product is in the final stage of development and Siemens Healthineers launched it last year in Europe. They are collecting feedback and rolling it out to more EU countries. At the same time, they continue to make improvements to the device and add features.
While the device currently only has the high-sensitivity troponin test for heart attacks, this is only the beginning. Preparing for the long-term vision, the team has already designed and built a feature into its device called multiplexing so multiple tests can be processed on one disposable insert.
In the near future, the team will set its sights on other markets outside Europe. After all, the goal is that this next-generation point-of-care testing device becomes the industry standard for diagnostics, creating better patient care across the world.
And because of its interesting history of entrepreneurship, working with tech giants, and cutting-edge tech development in Eindhoven, it won’t just be a win for Siemens Healthineers, but also a win for the Brainport region.
